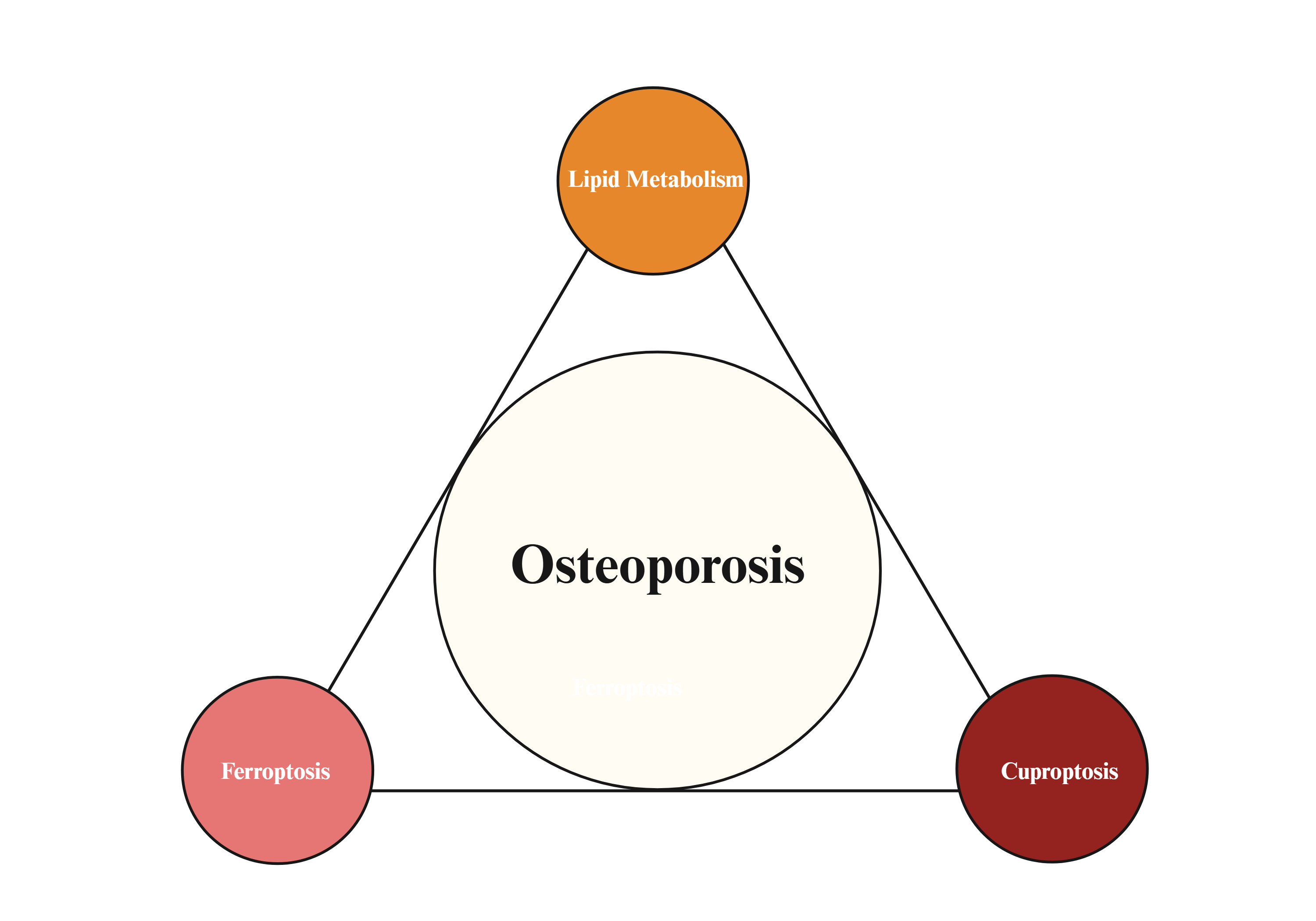
Cellular mechanisms of osteoporosis: A comprehensive perspective on ferroptosis, cuproptosis and lipid metabolism abnormalities
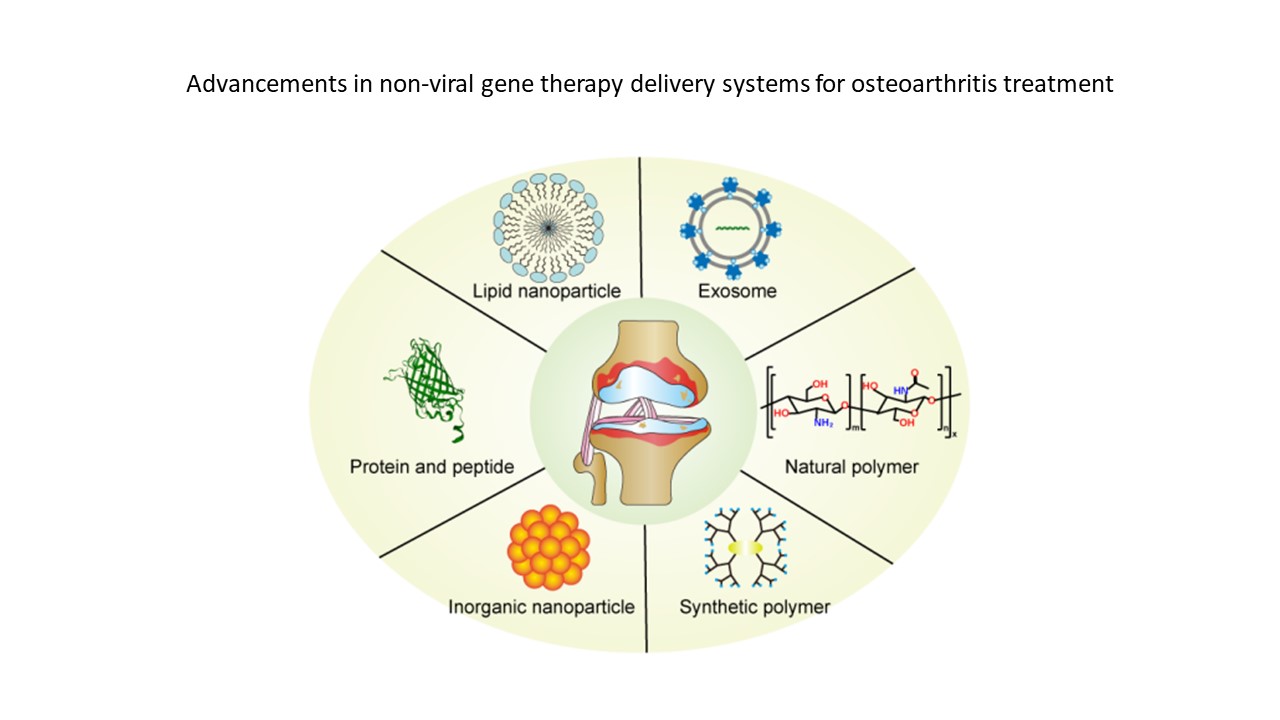
Non-viral gene delivery systems for osteoarthritis therapy
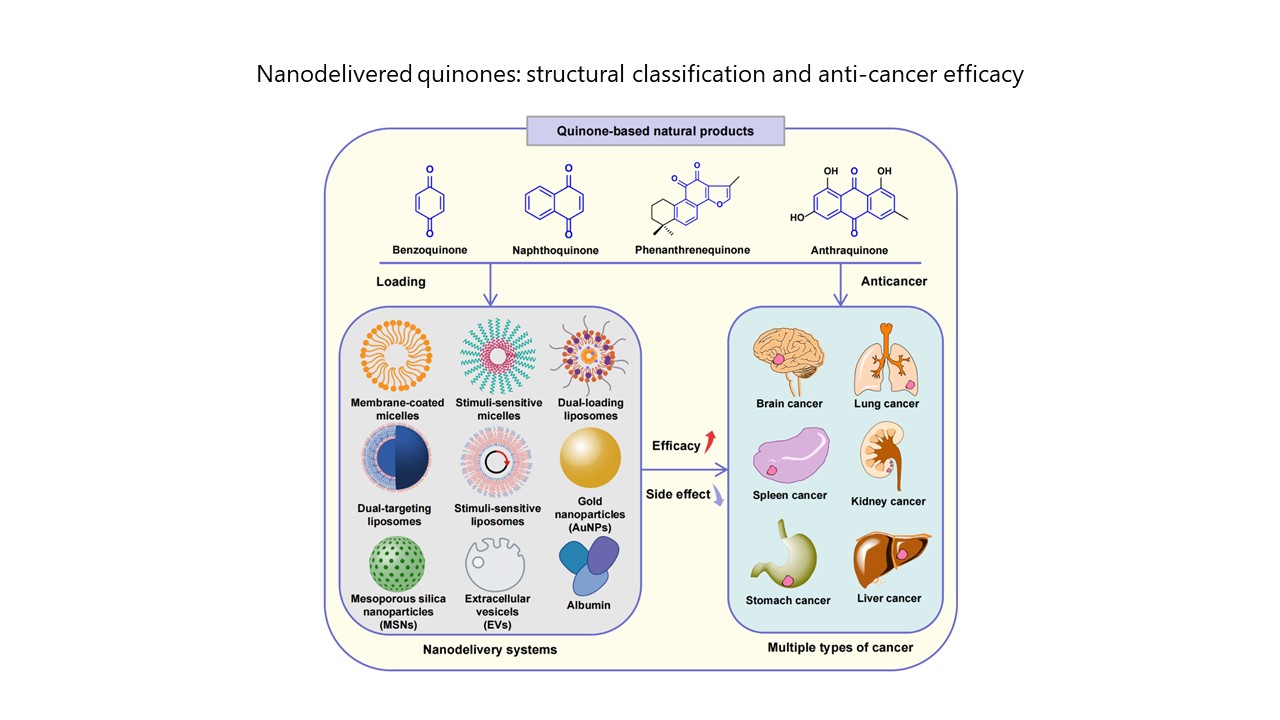
Empowering of novel anti-tumor formulations with quinone-based active natural products
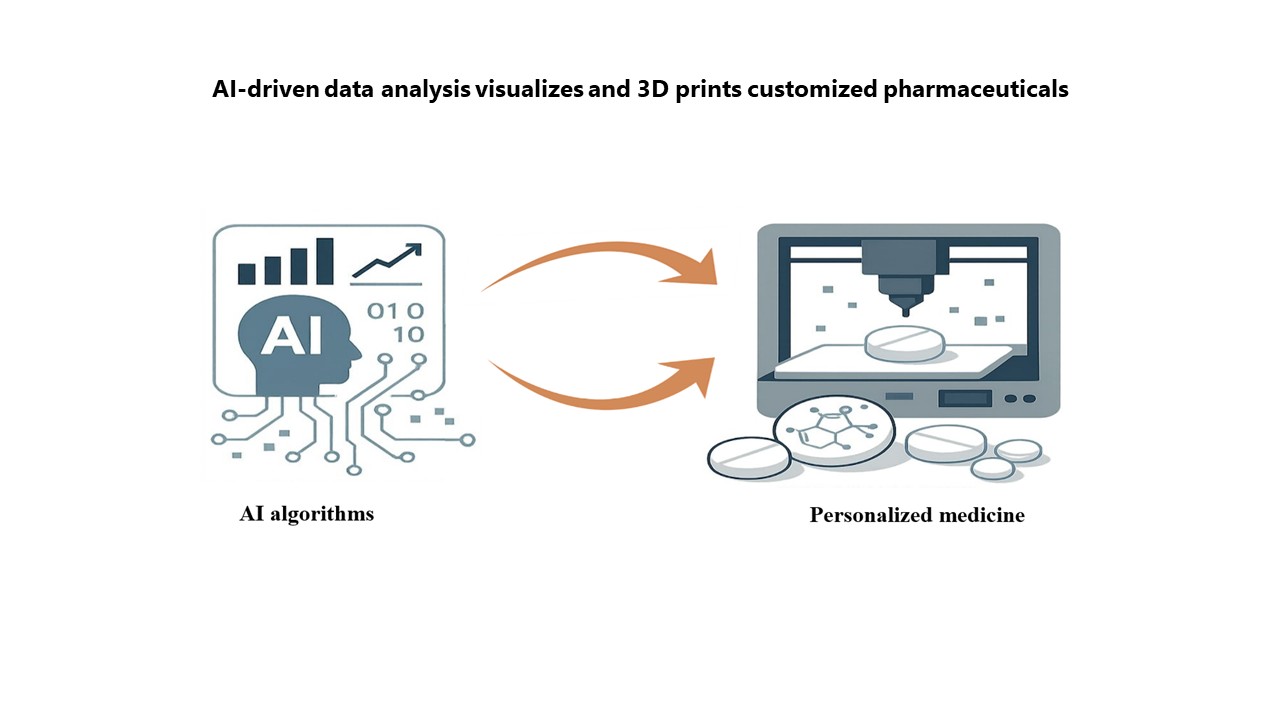
Building the next frontier: Artificial intelligence in 3D-printed medicines
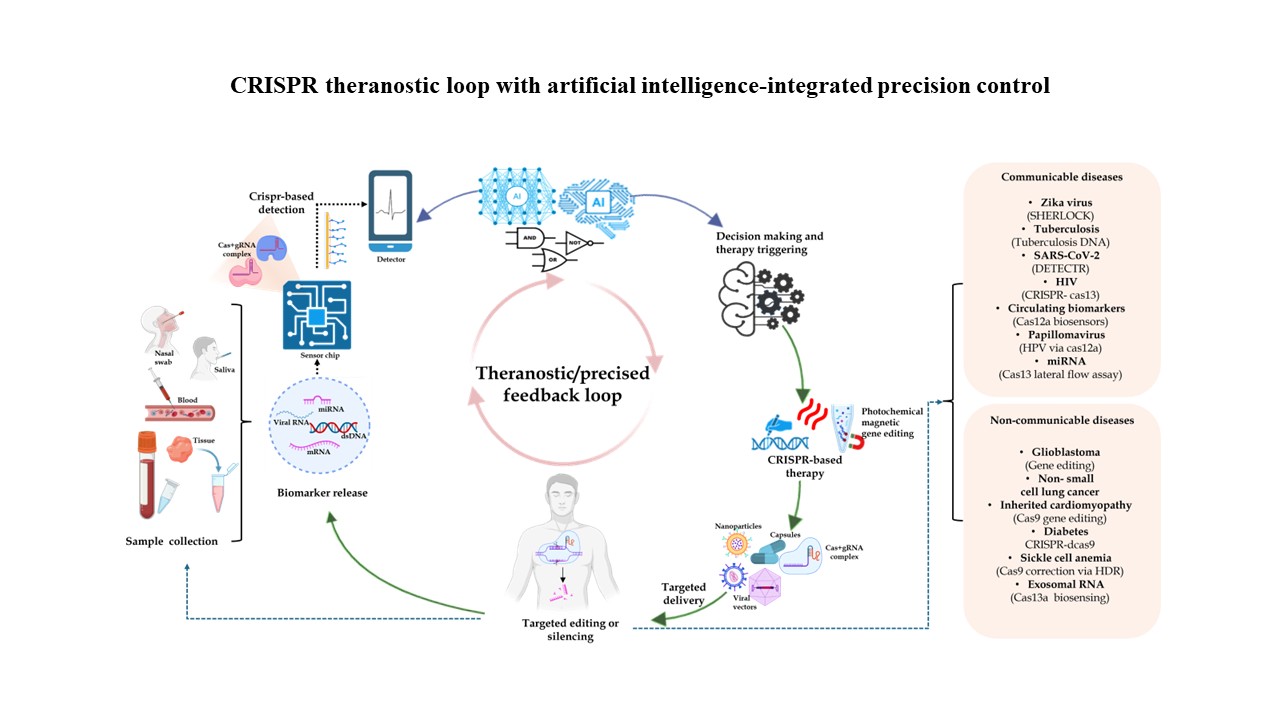
An overview of CRISPR-artificial intelligence theranostics: Current and emerging applications
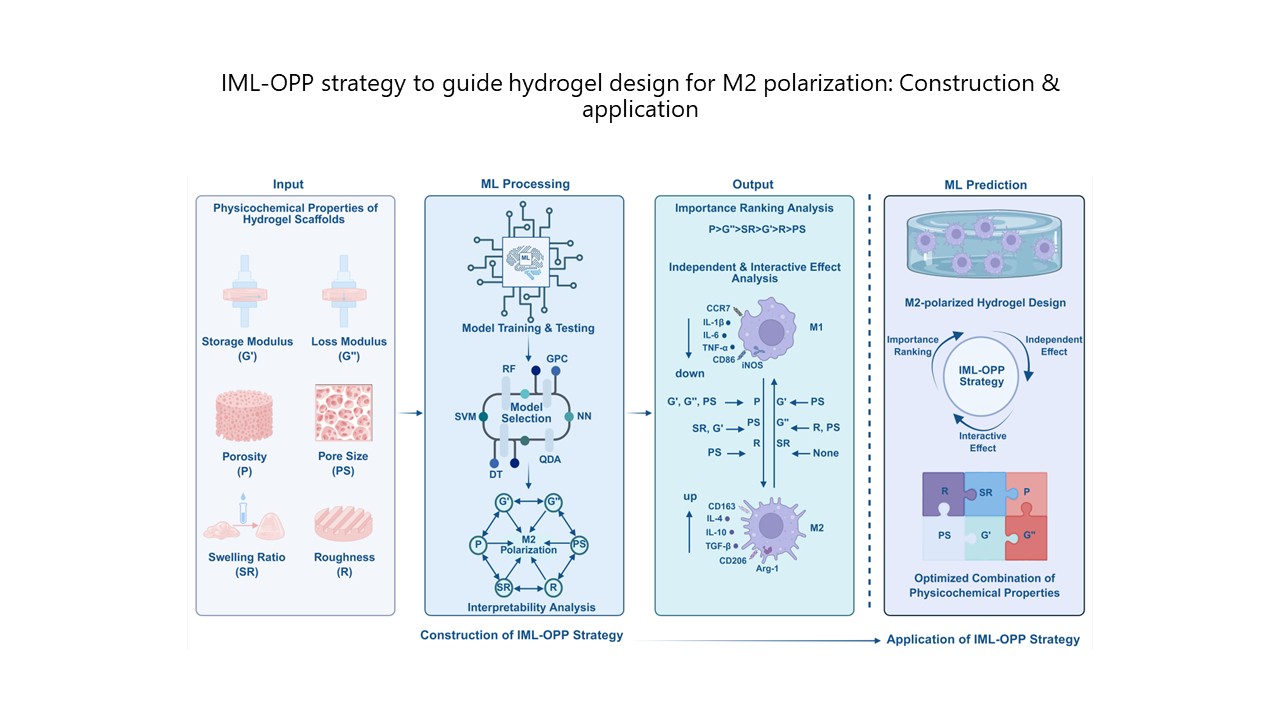
Interpretable machine learning-driven optimization of physicochemical properties in hydrogel scaffolds to promote macrophage polarization
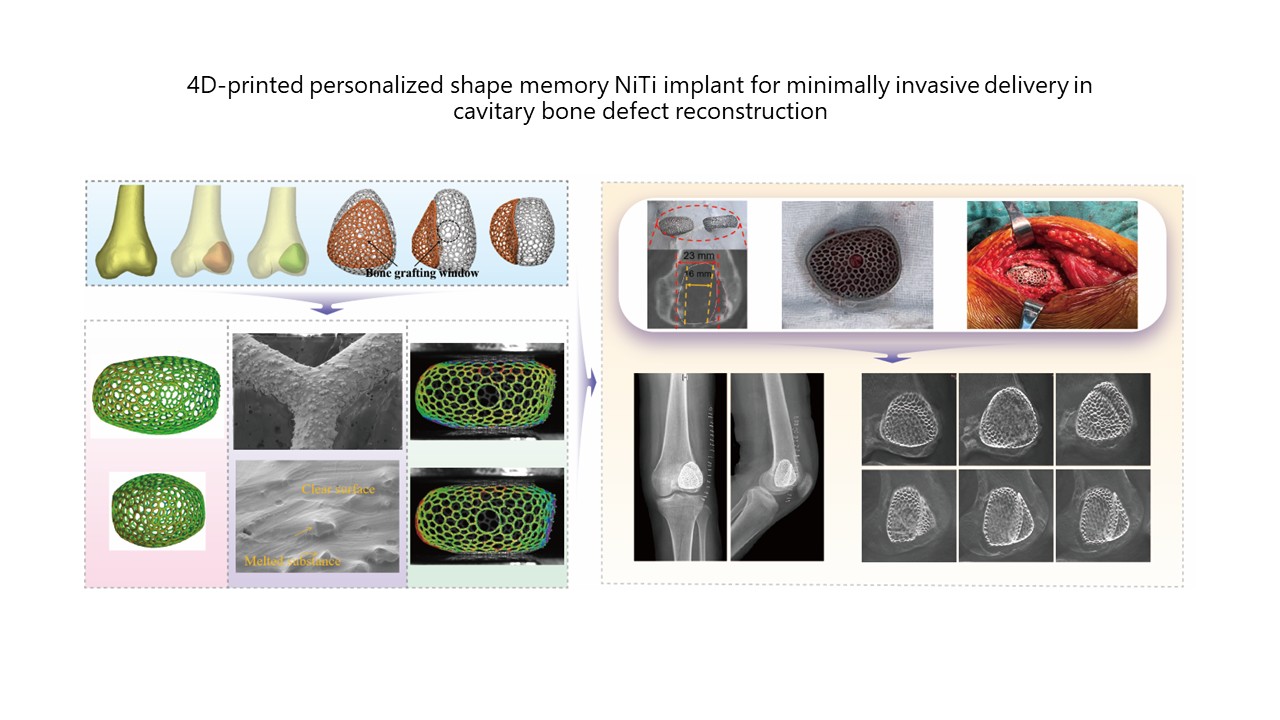
Four-dimensional-printed personalized shape memory NiTi implant for minimally invasive delivery in cavitary bone defect reconstruction
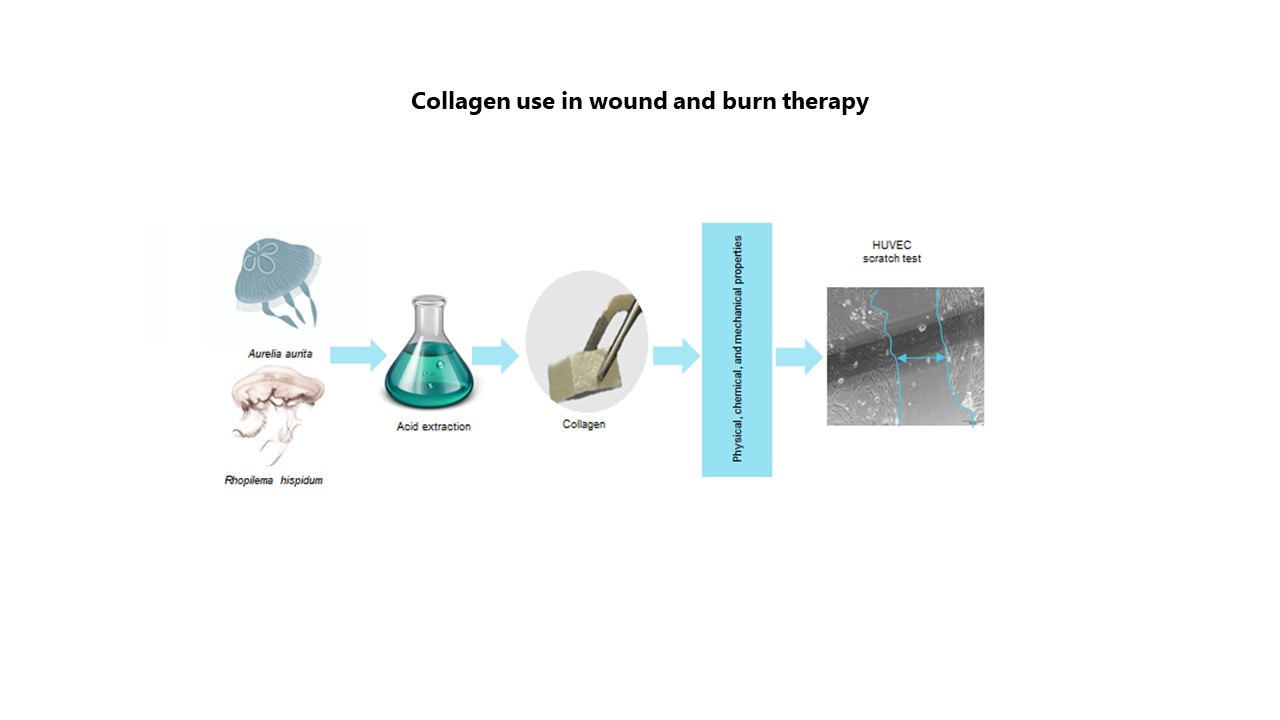
Comparative analysis of collagen from different sources for wound and burn management
Advances in bone defect repair using bio-3D printing technology: Innovations and challenges in mechanically assisted post-bioprinting strategies
Smart biomaterials in regenerative medicine



